Anna-Lena Brecher and Lena Kraft, Hannover Medical School; Linea Schmidt, University of Potsdam, Icahn School of Medicine at Mount Sinai, and German Society of Digital Medicine; Anne Sophie Platzbecker, University of Potsdam, German Society of Digital Medicine, Dresden University of Technology, Medical University Lausitz – Carl Thiem; Ariel D. Stern, University of Potsdam and Icahn School of Medicine at Mount Sinai; and Volker E. Amelung, Hannover Medical School and Private Institute for Applied Health Services Research
Contact: amelung.volker@mh-hannover.de
Abstract
What is the message? In 2019, Germany introduced the first formal process to authorize and reimburse digital health applications that demonstrably add value to patient care and the overall care delivery infrastructure. While the core idea is compelling, its implementation in practice has revealed challenges – highlighting the need for value-based payment, enhanced integration into patient journeys, and targeted strategies to engage providers, payers, and patients.
What is the evidence? The article draws on policy documents, implementation reports, and expert commentaries published since the launch of reimbursable digital health applications. It synthesizes these insights to identify practical barriers and strategic enablers, offering a forward-looking perspective grounded in the real-world experience from the last five years.
Timeline: Submitted: May 31, 2025; accepted after review: February 2, 2026.
Cite as: Anna-Lena Brecher, Lena Kraft, Linea Schmidt, Anne Sophie Platzbecker, Ariel D. Stern, Volker E. Amelung. 2026. Built to Run, but Stuck in First Gear? Lessons Learned and the Road Ahead for Digital Health Applications 2.0. Health Management, Policy and Innovation (www.HMPI.org). Volume 11, Issue 1.
Digitizing Health – Between Policy Vision and System Reality
The U.S, Food and Drug Administration (FDA) was among the first regulatory authorities to systematically address the evaluation and approval of digital health applications. Initial guidance for assessing mobile medical applications was published in 2011, with a finalized version released in 2013. This regulatory framework set the stage for the approval of innovative digital therapies that, in terms of efficacy, are comparable to conventional medicines [1]. Such tools are now commonly referred to as “Digital Therapeutics,” or “DTx,” and are defined as “evidence-based therapeutic interventions driven by software to prevent, manage, or treat a medical disorder or disease” [2]. A notable example is the reSET app from Pear Therapeutics, cleared by the FDA in 2017 as the first DTx using cognitive behavioral therapy for the treatment of substance use disorder [3].
In 2020, Germany introduced its own comprehensive strategy for digital health applications (Digitale Gesundheitsanwendungen, or “DiGA”) following the German Parliament’s passage of the Digital Healthcare Act on November 7, 2019. This legislation established Germany as the first country worldwide to formally reimburse physician-prescribed digital health applications under its mandatory, statutory health insurance framework.
Germany’s equivalent of the FDA, the Federal Institute for Drugs and Medical Devices ((Bundesinstitut für Arzneimittel und Medizinprodukte, or BfArM), does not have the same influence on the medical device market as its U.S. counterpart. The BfArM does not determine which applications are approved for distribution in the medical device sector or which applications are validated as medical devices; these decisions are made by a decentralized group of European Notified Bodies, independent organizations responsible for product review.
Furthermore, unlike in the United States, where regulatory and reimbursement decisions are based on separate processes, Germany’s digital health applications, or DiGA, are reimbursed by health insurers. Jens Spahn, Federal Minister of Health from 2018 to 2021, emphasized the importance of a systematic testing process to ensure the quality of digital applications, underlining that the system should reimburse only those tools demonstrating evidence of clinical value. This approach was designed to bolster Germany’s digital healthcare market while ensuring the quality of care. Indeed, the guiding philosophy behind the launch and subsequent growth of DiGA was to support both the digitalization of the German healthcare system and the improvement of care. The enactment of the Digital Healthcare Act provided a functioning regulatory framework within a matter of months, establishing a fast-track process for evaluating DiGA, a collaboration between the application manufacturer and BfArM; provisional approval to facilitate a trial evaluation, and a pathway to price negotiations (see Figure 1).
Moreover, the BfArM emphasizes the importance of a systematic approach based on rigorous evidence for identifying applications that are fit for incorporation into standard care. The BfArM’s fast-track process tests applications for safety, functional suitability, data protection, and proven positive effects on care [4]. Beyond regulatory approval and pricing, the true test for reimbursable digital health applications lies in their real-world applicability within Germany’s multifaceted care structures. A core component of the DiGA system is its seamless integration into existing care infrastructure: applications are not intended to be standalone solutions, but to complement and enhance existing treatment processes.
At first glance, around six years after the passage of the DVG and over five years after the launch of the first DiGA, it may seem like this system is working. While other countries such as the United States are still searching for suitable authorization and reimbursement mechanisms for digital health applications, Germany has established an innovative, legally enshrined procedure that systematically evaluates and integrates digital applications into the standard of care. Indeed, the French PECAN system, which was launched in March of 2024, borrows much of its design from the DiGA fast-track process. Other European countries like Belgium are in the process of testing similar digital therapeutic reimbursement pathways.
However, the reality is much more complex. On the one hand, the provisional authorization of digital health applications for an initial period of one year continues to represent a logical approach: it allows rapid access to therapy and provides manufacturers time to provide the necessary evidence. In practice, however, many manufacturers, particularly those with limited financial resources, are burdened by the “double regulation” and additional bureaucracy associated with submissions for both provisional approval and then permanent approval of a new application. This raises fundamental questions about the practicality and sustainability of a process that intends to be start-up-friendly.
Concurrently, the DiGA experience to date establishes an important foundation for system improvements, among them: supporting manufacturers with more specialized assistance in generating real-world evidence outside of traditional clinical trials and using data such as medical claims, electronic health records, and app metadata; establishing a more robust connection to care pathways; and adopting a more timely approach toward realistic pricing models. The fast-track process could be expanded further, for example through graduated approval models currently used for classifying medical devices. These steps would improve the regulatory fit between application risk, evidence, and healthcare relevance.
To maintain momentum and avoid stagnation, the framework for reimbursable digital health applications needs to transition from a product-centric focus to that of an evolving system. The next phase, “DiGA 2.0,” must therefore concentrate on enhancing problem-solving capabilities, ensuring usability in low-threshold care settings, and providing transitional support until the product is integrated into the market – all while ensuring that the German insurance system continues to reimburse only those tools that truly create value for patients and the healthcare system overall. This article examines how reimbursable digital health applications can be further developed to align with the overarching goal of patient-centered care.
Legal Framework and Political Course Setting
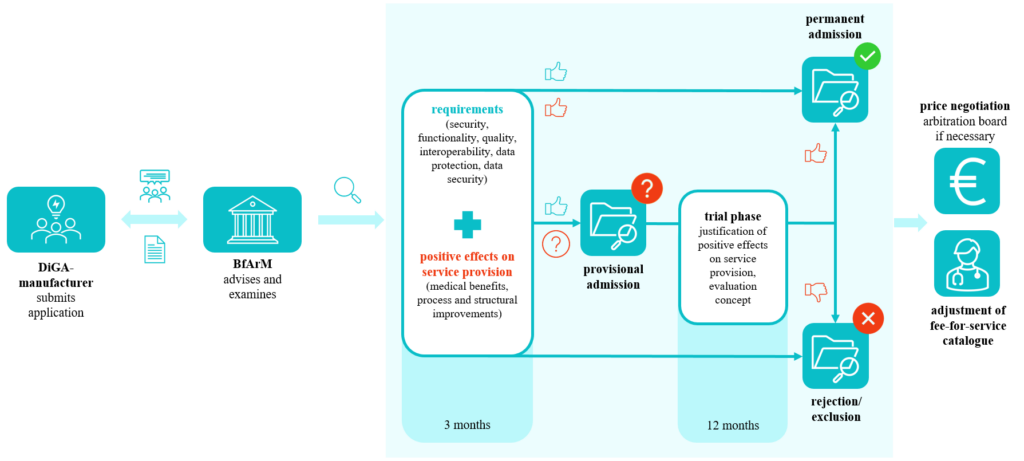
DiGA are risk class I, IIa or IIb medical devices (as defined by the EU Medical Device Regulation) that are primarily based on digital technology and which carry the European Conformity safety certification. Review and listing follows the fast-track process, the only option for risk class I or IIa medical devices, and takes three months from the time manufacturers submit their application. Following the same evidence requirements, risk class IIb medical devices are eligible only for permanent DiGA listing, without the fast-track process. In addition to information such as data protection or product characteristics, the fast-track process (see Figure 1) requires the respective manufacturers to submit quantitative comparative studies that provide evidence of the DiGA’s positive health effect.
Figure 1: The DiGA Fast-Track Process
Note: This figure only holds for medical devices of risk class I and IIa. Source: Own illustration based on [4]
Following receipt of a complete application, the BfArM will verify within three months whether the digital health application meets the required criteria. If the evidence is incomplete, the application can be provisionally listed in the DiGA directory for up to 12 months – with the possibility for a trial period extension of up to two years – while the manufacturer provides the necessary studies. If the manufacturer can demonstrate so-called positive care effects within this period, the DiGA will be permanently listed. If the required evidence is not provided within this period, it will be removed from the DiGA directory’s approved list [4, 5].
While this approach offers DiGA a structured entry into the healthcare system, it also exposes tensions between early-market access and insufficient medium-term evidence. As market access is often the main concern for digital and other products (rather than market exit in the event of failure), systematically removing applications that cannot survive on the market or do not meet requirements after the review process has been completed, represents a practical challenge. Yet doing so is of the utmost importance to the integrity of the DiGA system, ensuring that no applications remain on the market if they lack evidence of clear benefit.
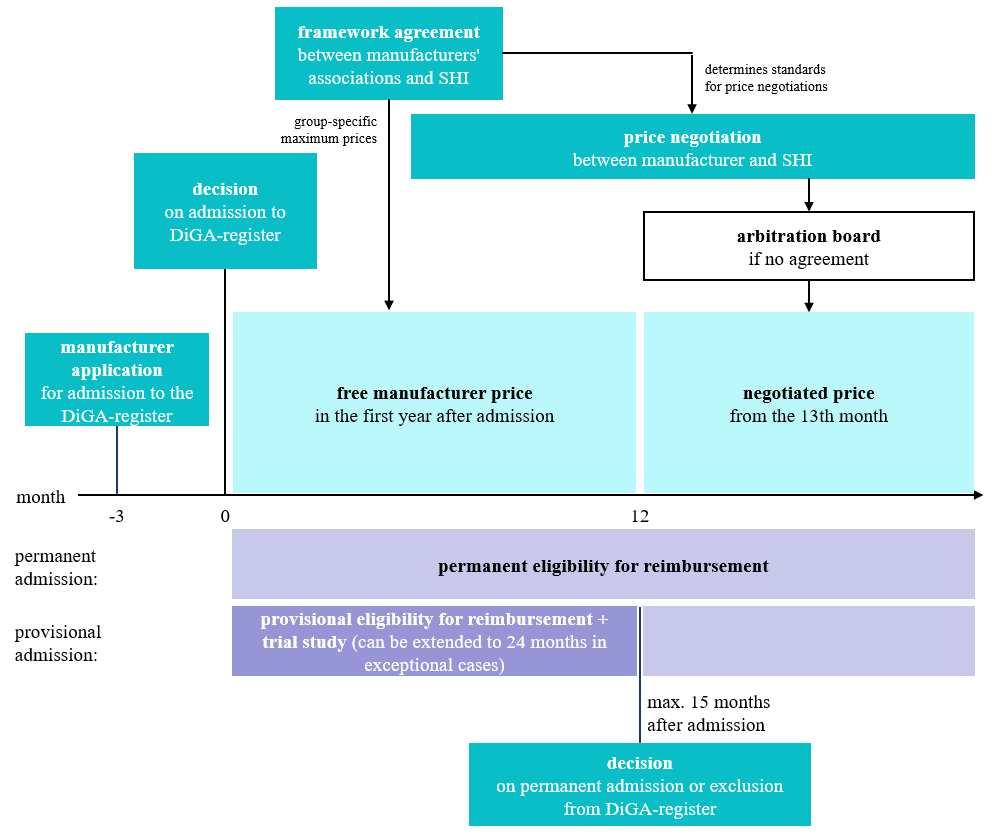
The DiGA fast-track process has been hailed as a breakthrough both nationally and internationally, particularly because of the accelerated approach and the low-risk classification specific to DiGA [6]. The determination and negotiation of DiGA pricing follows a clearly delineated process (see Figure 2). During the first year that an application is included in the DiGA directory, the provisional manufacturer’s price applies. The renegotiated reimbursement amount takes effect starting the 13th month. The subsequent permanent price is renegotiated between the manufacturers and the National Association of Statutory Health Insurance Funds (GKV-Spitzenverband). The BfArM informs the GKV-Spitzenverband about the price negotiation to be included in the directory [4].
Figure 2: Process of price determination and negotiation for DiGA
Source: Own illustration based on [7]
From Theory to Practice
Germany’s DiGA framework is entering its sixth year, allowing for an initial assessment of progress to date and starting points for discussion about the system’s further development. Some regulations were updated recently and are not reflected in the past data [8].
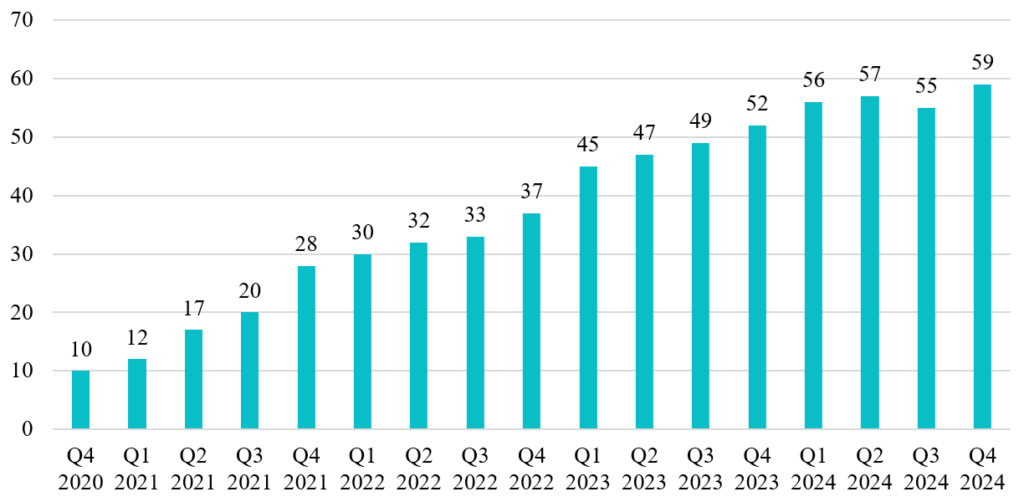
Figure 3: Evolution of the number of listed DiGA, as of October 31, 2025
Source: Own illustration based on [8]
From the initial inclusion of the first digital health application in the directory in September 2020 to December 31, 2024, a total of 861,000 DiGA prescriptions were filled by insured persons at least once. The number of activation codes redeemed per quarter (a common measure of utilization employed in the German healthcare system) has risen steadily. However, there are considerable DiGA utilization disparities. Concurrently, the number of listed DiGA has also increased steadily over time, growing from 10 DiGA in the fourth quarter of 2020 to 56 DiGA at the beginning of 2024. Since then, the number of DiGA in the directory has plateaued (see Fig. 3).
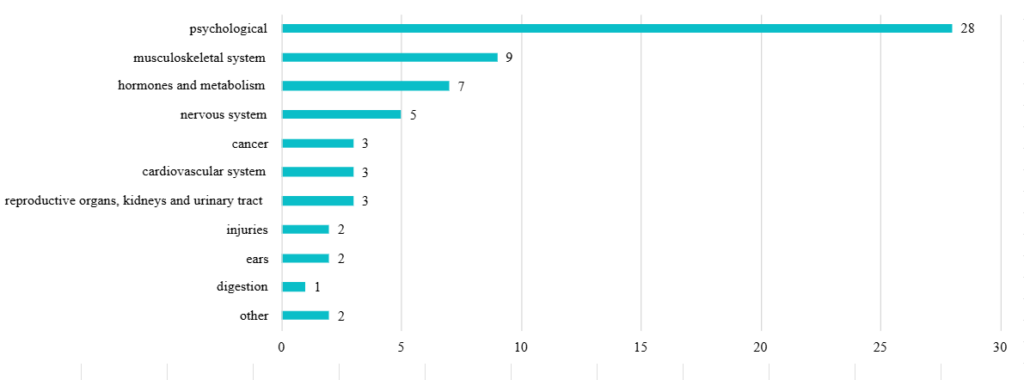
The majority of listed DiGA (51%) relate to mental and behavioral health (see Fig. 4). In second place, with around 12% of approved DiGA, are applications for musculoskeletal disorders, followed by metabolic diseases with around 11%. Seven other indication areas are also covered [8].
Figure 4: Applications listed in the DiGA-directory by indication area, as of October 31, 2025
Note: Assignment to multiple categories possible. Source: Own illustration based on [9]
A total of 73 digital health applications have been listed in the DiGA directory since its inception. Of these, 47 applications (64%) were permanently included after the manufacturers demonstrated positive care effects; 16 digital health apps have been removed from the directory due to an inability to prove benefit. The remaining 10 digital health applications are currently listed provisionally while trials are conducted to prove their benefit to patients and care processes. According to the GKV-Spitzenverband, only every second DiGA that was provisionally included in the health insurance catalogue for testing, achieved the promised benefit over the period of up to 24 months [8].
In the first year after launch, the price of a DiGA is set by its manufacturer, considering various costs associated with R&D such as development or clinical trials, and are capped by a maximum amount regulation. The manufacturers’ quarterly prices (90-day course of treatment) currently range from 119 to 952 euros at launch (approx. US $140 to US $1,121). In 2020, the average price was still 411 euros (approx. US $484), by 2024 it had increased to nearly 500 euros (approx. US $589). Following the permanent inclusion of DiGA in the directory, its manufacturer and the GKV-Spitzenverband negotiate the price that will apply beginning with the 13th month. The average negotiated price per 90-day course of treatment is 266 euros (approx. US $313), and negotiated prices range from 189 to 248 euros per quarter (approx. US $223 to US $292) [10].
Barriers and Bottlenecks
Despite Germany’s clear framework for the authorization and reimbursement of evidence-based digital health applications, stakeholder acceptance has been a challenge. In recent years, numerous studies have shown that some physicians are skeptical about digital therapeutics and this new approach to healthcare delivery. Primary areas of concern are the scientific evidence for effectiveness, the potential DiGA-related increase in physician workload, uncertainties regarding legal conditions and data protection, and the view that the remuneration of clinician services is insufficient compared to the perceived high costs of digital health applications [10, 11].
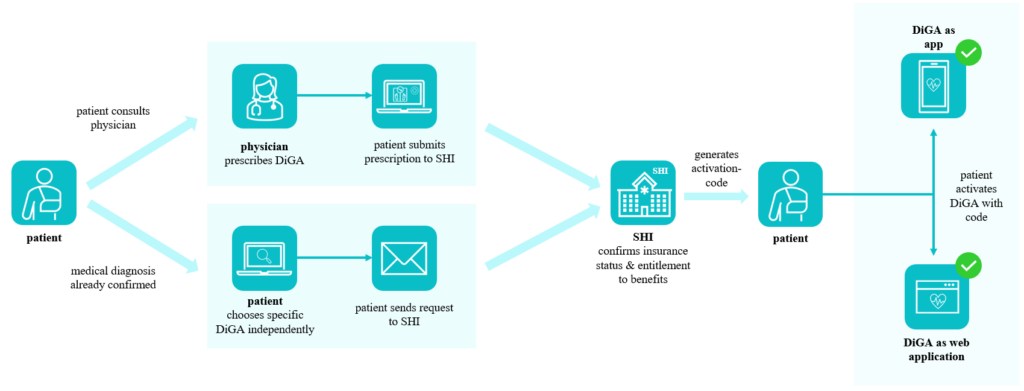
Figure 5: Pathways for patients to access a DiGA
Source: Own illustration based on [12]
Potentially the biggest barrier, even among doctors who generally agree with the DiGA concept, is a lack of knowledge about which DiGA are available, how they can be prescribed, and for which patients they are suitable. In many respects, this should not surprise us: Germany has the oldest physician population in Europe, many of whom are not digital natives [13]. Furthermore, the absence of physician digital education during medical training and its ongoing absence as a requirement in continuing medical education, points to a persistent digital literacy gap. In practice, only a small proportion of doctors regularly prescribe DiGA and plan to do so in the future [11, 14–16].
There are two different pathways to accessing and using DiGA. In addition to a physician prescribing digital health applications, patients may research and select suitable DiGA for their conditions and apply for coverage from their insurance provider directly (see Fig. 5). However, this access option is only used in around 10% of cases, highlighting the importance of doctors, especially GPs, in recommending DiGA to patients and being knowledgeable and supportive of digital therapeutics [8]. Patients who request an activation code from their health insurance company to gain access to the DiGA also encounter bureaucratic hurdles. Often, too much time passes, and ultimately, 17% do not redeem their code at all [17].
Insurers have also not wholeheartedly accepted DiGA. Above all, they have criticized what are perceived as high prices, particularly those set by the manufacturers during the first 12 months. That criticism has at times been linked to the view that reimbursement of provisionally approved DiGA equates to a subsidy for manufacturers. This is particularly salient for insurers in cases where the manufacturer fails to demonstrate positive effects through a study conducted within the trial period, resulting in the application’s removal from the DiGA directory. As the number of activated prescriptions continues to increase (+ 85% in 2024), insurers’ DiGA expenditures will grow proportionally. Germany’s insurance industry association therefore regularly questions the extent to which DiGA expenditure actually contributes to an improvement in healthcare [8].
Conversely, DiGA manufacturers have expressed discontent with the high level of bureaucracy involved in applying to the BfArM for inclusion in the DiGA directory, citing numerous regulatory and certification requirements as contributing factors to high (and growing) development costs. When it comes to generating evidence of the benefits associated with a DiGA, the question arises whether randomized controlled trials should always be the method of choice. Although the DiGA fast-track process explicitly allows for evidence generation beyond traditional randomized controlled trials, in practice, most manufacturers still rely on these trials due to perceived regulatory uncertainty and a lack of successful precedent for real-world evidence [18].
Lessons Learned and the Way Forward
Germany was the first country worldwide to implement a structured process for the assessment and reimbursement of digital health applications. In doing so, it positioned itself as a pioneer in this field. Other countries, such as France, Austria, and Belgium, have since followed, establishing their own frameworks to integrate digital therapeutics into standard care [17]. Germany’s DiGA pathway and the first half decade of experience have shown that the concept of reimbursable digital health applications could be improved and accelerated with targeted changes. For countries at the early stages of developing similar pathways, the German experience offers both inspiration and critical lessons. The core idea of selecting high-quality, evidence-based digital tools and embedding them into routine care has great potential. However, certain aspects of the system’s operationalization and execution could be improved to enhance acceptance among healthcare providers, reduce the perceived financial burden on the public healthcare system, stimulate innovation, and improve health outcomes for patients.
Improved Integration into Patient Journeys
All stakeholders in the healthcare system, especially in a system as strongly characterized by self-administration as in Germany, should be involved in the implementation of new processes early on. Together with insurers and healthcare providers, key therapeutic needs should be identified where digital health applications can offer real added value. Mental and behavioral health, for example, can accommodate easily digitizable approaches (such as cognitive behavioral therapy) and experience a shortage of healthcare providers.
This also involves considering how such health apps can be better integrated into existing care processes so that they are not perceived as foreign bodies within the system, but rather as an integral part of the patient’s journey. A recent example could be the complementarities between DiGA and GLP-1 agonist drugs like Semaglutide or Tirzepatide, which require a comprehensive therapeutic approach to achieve sustainable positive outcomes, and digital applications that support patients in monitoring, education, and implementing necessary lifestyle changes. By embedding such digital tools into the care pathway alongside pharmacological treatments, the overall effectiveness of therapy can be enhanced, adherence improved, and patients empowered to take an active role in managing their health. This kind of integration not only strengthens the continuity of care, but also underscores the added value of digital health solutions, not just as a product, but a real component of more holistic and patient-centered care and solutions [19].
Adjustment of Remuneration and Pricing
To ensure that physicians are motivated to familiarize themselves with prescribable health apps and provide targeted guidance to patients – including onboarding and ongoing support – it is essential to establish appropriate remuneration for these services. At the same time, the pricing of reimbursable digital health applications must be carefully balanced. Pricing should provide manufacturers with sufficient incentives to pursue the fast-track process, while avoiding undue strain on insurers and preserving stakeholder acceptance. Currently, all DiGA in Germany are reimbursed at a flat rate per quarter, regardless of usage or impact. In the future, more flexible, value-based reimbursement models should be considered to better reflect the actual benefit delivered. Additionally, introducing moderate patient co-payments – similar to those for pharmaceuticals – could contribute to the financial sustainability of these applications without creating significant access barriers [19].
From a health system perspective, the sustainability of DiGA reimbursement will critically depend on whether they can demonstrate value in terms of outcomes achieved relative to costs. The planned integration of post-market performance assessment (in Germany known as “Anwendungsbegleitende Erfolgsmessung”) is one step into this direction. Value-based healthcare concepts should guide this next phase of DiGA development, pricing and evaluation. What has been explicitly discussed is an outcome-based remuneration model with 20% reimbursement based on performance, which potentially improves the incentives alignment between manufacturers, payers, and providers. For that, it will be vital to select appropriate success measures that enable comparisons across different digital applications while also accounting for differences in indication areas, targeted outcomes, and intervention characteristics. Transparent post-market performance assessment is also relevant for research to better understand digital therapeutics as a care model, contributing to a broader availability of real-world evidence.
Flexible Approaches for Evaluating Digital Health Applications
To demonstrate the benefits of digital health applications, different evaluation methods are required than those used in clinical trials for new drugs. A range of additional study design aspects must also be considered to adequately reflect the characteristics of digital interventions. This includes the definition and establishment of novel clinical and non-clinical endpoints that may diverge from conventional measures. In addition to objective clinical outcomes, patient-centered dimensions such as quality of life, symptom perception, mental well-being, empowerment, self-management, and treatment adherence should be incorporated.
Further, many experts believe it will be key to adapt Patient-Reported Outcome Measures (PROMs) and Patient-Reported Experience Measures (PREMs) to digital contexts and establishing methodological guidance for their integration into digital platforms. These capture dimensions such as quality of life, symptom perception, mental well-being, and empowerment in self-management – all factors explicitly the DiGA regulation recognizes as “positive care effects.” DTx are particularly suited to continuously collect such outcomes through in-app surveys, wearable integration, or sensor-based monitoring. These types of endpoints not only allow for real-time care adjustment, but also provide real-world evidence that is highly relevant for patients themselves. Embedding PROs and PREMs into routine DiGA evaluations would strengthen patient-centeredness and create an evidence base that goes beyond short-term randomized clinical trials, reflecting real-world effectiveness, patient-centric endpoints, and adherence [18]. In line with this shift toward pragmatic evidence generation, the FDA announced in December 2025 that it would adopt a more flexible approach toward real-world evidence, allowing certain medical device submissions without requiring identifiable individual patient-level data [20].
In addition, opportunities to use meta-data from within the DiGA themselves, like usage duration and frequency, may create additional opportunities to understand, predict, and support digital therapeutics adherence. This data processing and potential secondary use for research purposes is subject to the data-protection and IT-security requirements set by Germany’s Federal Office for Information Security, outlined in § 4 DiGAV.
Effective Communication and Education
Above all, effective communication is essential. Germany’s case clearly shows that meaningful and well-designed innovations do not diffuse on their own, especially given the limited time and resources of everyday medical care. What is needed are persuasive communication strategies, transparent and accessible information, and targeted efforts to enhance digital health literacy among both healthcare providers and patients.
A critical yet often underestimated success factor is continuing education. To address DiGA knowledge gaps among physicians and nurses, structured training formats should be integrated into medical and nursing curricula as well as continuing education programs. In particular, new roles such as Advanced Practice Nurses or Health Community Nurses could play a pivotal role in onboarding patients, conducting home visits, and managing telemonitoring processes. By equipping healthcare professionals with digital competencies and creating new career paths that explicitly include digital health responsibilities, DiGA use can be embedded more effectively and sustainably into routine care.
Data Interoperability and Equity
A major limitation for scaling DiGA remains the lack of interoperability with existing electronic health record systems and practice software. Without seamless data exchange, physicians are unlikely to integrate digital applications into their workflows. National initiatives for standardized APIs and interoperable data infrastructures therefore represent a vital prerequisite for “DiGA 2.0.” At the same time, equity concerns must be addressed. Current uptake shows strong variation by age, socioeconomic status, and region. Especially in rural areas or among older patients, limited digital literacy and infrastructure barriers pose exclusion risks. Targeted support measures, simple user interfaces, and low-threshold onboarding services are essential to ensure that digital therapeutics contribute to reducing, rather than exacerbating, healthcare inequalities.
Conclusion
Germany’s experience with the DiGA fast-track process illustrates the feasibility and challenges of integrating DTx into statutory healthcare. Critical enablers for success include meaningful integration into patient journeys, appropriate remuneration and pricing mechanisms, flexible and patient-centered evaluation approaches, effective communication and education, and robust data interoperability. Addressing these dimensions is essential to strengthening acceptance among healthcare professionals, payers, and patients and unlocking the full potential of digital health applications to improve health outcomes and system efficiency.
References
- Food and Drug Administration. Device Software Functions Including Mobile Medical Applications 2022. Available at: https://www.fda.gov/medical-devices/digital-health-center-excellence/device-software-functions-including-mobile-medical-applications?utm Accessed May 26, 2025.
- Kampouraki D. Digital Therapeutics (DTx). Available at: https://www.edps.europa.eu/press-publications/publications/techsonar/digital-therapeutics-dtx_en Accessed November 03, 2025.
- Zagorski N. FDA Clears First Prescription-Based ‘Digital Therapeutic’. Psychiatric News 2017;52(23):1.
- Bundesinstitut für Arzneimittel und Medizinprodukte. Das Fast-Track-Verfahren für digitale Gesundheitsanwendungen (DiGA) nach § 139e SGB V: Ein Leitfaden für Hersteller, Leistungserbringer und Anwender. Bonn 2020.
- Bertelsmann Stiftung. Digitale Gesundheitsanwendungen und Patient-Reported Outcome Measures: Markt- und Potenzialanalyse 2022.
- Stern AD, Matthies H, Hagen J, et al. Want to See the Future of Digital Health Tools? Look to Germany. 2020. Available at: https://hbr.org/2020/12/want-to-see-the-future-of-digital-health-tools-look-to-germany Accessed November 03, 2025.
- Techniker Krankenkasse. DiGA-Report 2022 2022.
- GKV-Spitzenverband. DiGA-Bericht des GKV-Spitzenverbandes – 2024: Bericht über die Inanspruchnahme und Entwicklung der Versorgung mit Digitalen Gesundheitsanwendungen 2025.
- Bundesinstitut für Arzneimittel und Medizinprodukte. DiGA-Verzeichnis 2025. Available at: https://diga.bfarm.de/de/verzeichnis Accessed May 01, 2025.
- Weik L, Fehring L, Mortsiefer A, et al. Understanding inherent influencing factors to digital health adoption in general practices through a mixed-methods NPJ Digit Med 2024;7(1):47. doi:10.1038/s41746-024-01049-0 [published Online First: 27 February 2024].
- Dahlhausen F, Zinner M, Bieske L, et al. Physicians’ Attitudes Toward Prescribable mHealth Apps and Implications for Adoption in Germany: Mixed Methods Study. JMIR Mhealth Uhealth 2021;9(11):e33012. doi:10.2196/33012 [published Online First: 23 November 2021].
- Bundesinstitut für Arzneimittel und Medizinprodukte. Informationen für Leistungserbringende o.J. Available at: https://diga.bfarm.de/de/leistungserbringende Accessed May 09, 2025.
- Medical workforce in the EU: an ageing profession 2015. Available at: https://ec.europa.eu/eurostat/web/products-eurostat-news/w/ddn-20250116-2 Accessed November 03, 2025.
- Brecher A-L, Herrgen J, Mueller T. DiGA in Primary Care and the Influence of Patient-Related Factors – Results of a Survey of Family Doctors. Stud Health Technol Inform 2024;313:55–61.
- Cirkel L, Lechner F, Schlicker N, et al. Adoption and perception of prescribable digital health applications (DiGA) and the advancing digitalization among German internal medicine physicians: a cross-sectional survey study. BMC Health Serv Res 2024;24(1):1353. doi:10.1186/s12913-024-11807-1 [published Online First: 6 November 2024].
- Dahlhausen F, Zinner M, Bieske L, et al. There’s an app for that, but nobody’s using it: Insights on improving patient access and adherence to digital therapeutics in Germany. Digit Health 2022;8:20552076221104672. doi:10.1177/20552076221104672 [published Online First: 3 July 2022].
- Spitzenverband Digitale Gesundheitsversorgung e.V. DiGA-Report 2024: Marktentwicklung digitaler Gesundheitsanwendungen 2025.
- Stern AD, Brönneke J, Debatin JF, et al. Advancing digital health applications: priorities for innovation in real-world evidence generation. Lancet Digit Health 2022;4(3):e200-e206.
- Spreafico AMC, Tarricone R, Stern AD. Comprehensive policies for scaling systemic and equitable integration of digital health technologies. NPJ Digit Med 2025;8(1):404. doi:10.1038/s41746-025-01820-x [published Online First: 4 July 2025].
- Food and Drug Administration. FDA Eliminates Major Barrier to Using Real-World Evidence in Drug and Device Application Reviews 2025. Available at: https://www.fda.gov/news-events/press-announcements/fda-eliminates-major-barrier-using-real-world-evidence-drug-and-device-application-reviews Accessed January 5, 2026.